
Moles count the number of atoms or molecules in a sample, while grams represent the weight of the sample. Because that’s such a large number, it’s almost always represented using scientific notation.  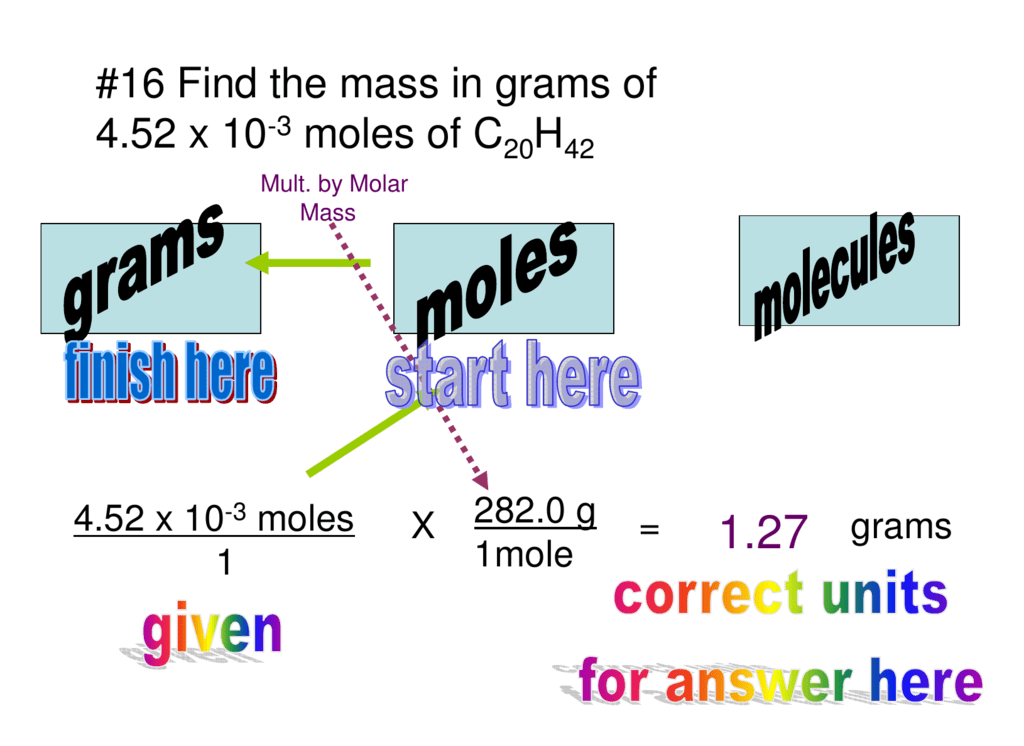
One mole is equal to 6.02214076 × 10 23 elementary units of matter, such as atoms or molecules. A gram is an SI unit of measurement for mass equal to 1/1,000 of a kilogram, and the mole is the SI base unit used to represent the quantity of a substance. Grams using the molar mass of of Fe 2O 3.Īll of these steps can be combined into a single equation as follows.Grams and moles are both units used in chemistry to measure matter in different ways. Obtain a factor that relates moles of Fe to moles of Fe 2O 3. Use coefficients from the balanced equation to Convert the mass of Fe to moles using the molar How much iron(III) oxide will be produced by the complete oxidation of 101 g of ironĪccording to the following equation? 4 Fe + 3 O 2 => 2 Fe 2O 3?įrom the question we deduce that Fe is the given substance and that Fe 2O 3 The ratio in which C 8H 18 and H 2O react is 18 moles H 2O for every 2 moles C 8H 18. Calculate the molecular weight of C 8H 18 (114 g/mol) and H 2O (18 g/mol). What weight of H 2O is produced by the reaction of 342 g of C 8H 18? Use the same logic as the first example. The only unit left uncancelled is grams O 2, the unit of oxygen. Apply the method of dimensional analysis. The 25 and the 2 are the stoichiometric coefficients of O 2 and C 8H 18, respectively.īecause 342 g of C 8H 18 is the only data actually stated, it becomes the starting point. The balanced equation indicates that C 8H 18 and O 2 react in the ratio of 25 moles of O 2 for every 2 moles of C 8H 18. Use the periodic table and the molecular formula to determine the molecular weights (O 2 = 32 g/mol and C 8H 18 = 114 g/mol). The question cites C 8H 18 and O 2, therefore, we need to calculate their molecular weights. Four tires are installed on each car (the appropriate ratio) and we can assume if one car and four tires arrive at the factory that a proper car will leave.)įirst we assemble all data and their factors. Because the atoms combine in simple whole number ratios, we can be assured that everything will react in the correct ratio if we weigh out the appropriate amount of chemicals. What weight of O 2 will react with 342 g of C 8H 18? (We can study this reaction two chemicals at a time. All units have been canceled except for grams of SO 2 (product). While reducing is not absolutely necessary (the ratio will cancel properly even if not reduced), a good chemistry student notices such things and will do it.ĥ. Notice that the ratio was reduced from 8 : 4 to 2 : 1 when placed in the factor-label form. This ratio comes from the coefficients in the balanced equation. Changes moles of FeS 2 (reactant) to moles of SO 2 (product). Convert grams of FeS 2 to moles of FeS 2.Ĥ. Write the mass information given in the problem.ģ. Write a balanced equation showing the formation of iron (III) oxide and sulfur dioxide.Ģ. If a furnace burns 100 g of FeS 2, how much SO 2 is produced?ġ. Iron pyrite, FeS 2, combines with oxygen from atmosphere to produce iron(III) oxide and sulfur dioxide. Convert moles of product to grams of product.Ħ. Convert moles of reactant to moles of product.ĥ. Convert mass of reactant to moles of reactant.Ĥ. Write the given mass on a factor-label form.ģ. Write a balanced equation for the reaction.Ģ. The question looks like this: "given this amount of reactant, how much product will form?"ġ. The most common type of stoichiometry calculation is a mass-mass problem. A quantitative study of chemical changes.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
